A review of the registered clinical trials of potential treatments for COVID-19 suggests that in many cases the trial design lacks features key to scientific value, such as the use of control groups and patient and clinician blinding.
You might also like:EMA Fast-Tracking Procedures for COVID-19 Medicines
The study from researchers at Johns Hopkins Bloomberg School of Public Health has been published in BMJ Open. The team analysed the 201 clinical trials for drugs or plasma that, as of 26 March, had been registered in the U.S. under ClinicalTrials.gov and in the international clinical trials registry maintained by the World Health Organization. It is noted that the number of registered trials in the list grew substantially from the beginning of March to 26 March, and as of 8 June there were over 2,000 entries.
Among the
201 trials analysed, half (100, 49.8%) were registered in China and over a
third (78, (37.8%) in the U.S.; 126 were recruiting participants from China, 31
from Europe, 14 from the U.S. The primary focus was to assess the effectiveness
of existing therapeutic products against acute COVID-19 disease. In total, the trials involved
92 drugs as well as antibody-containing blood plasma, including 64 in
monotherapy and 28 different combinations.
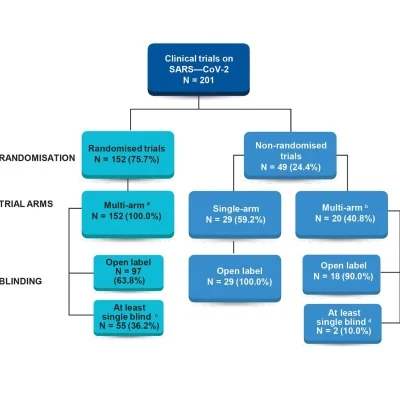
The researchers
found that most of these trials demonstrated design weaknesses. For example, one-third
of trials (67, 33.3%) lacked defined clinical endpoints (eg, discharge or
survival); nearly half sought to enrol fewer than 100 patients while 54 (26.9%)
fewer than 50 patients; about a quarter (49, 24.4%) did not have randomised
design; and nearly half (97, 48.3%) were open label. Therefore, the authors
said, many of these studies were “likely to yield only preliminary
evidence of a given treatment’s safety and effectiveness against COVID-19.” At
the same time they acknowledge the benefits of global registries for urgent
clinical trial research and as a platform for coordination and cooperation in
research.
Source: Johns
Hopkins Bloomberg School of Public Health
Image credit: Mehta HB et al. (2020) Characteristics of
registered clinical trials assessing treatments for COVID-19: a cross-sectional
analysis. BMJ Open,10:e039978. doi: 10.1136/bmjopen-2020-039978