LyGenesis, a biotechnology firm based in Pittsburgh, Pennsylvania, has embarked on a groundbreaking clinical trial involving an experimental treatment for liver failure. This innovative approach aims to grow an additional'miniature liver' within patients suffering from liver failure, offering a potential alternative to organ transplantation.
Alarming Impact of Liver Disease
Liver disease remains a significant health concern, with over 50,000 deaths attributed to it annually in the United States alone. In the advanced stages of the disease, accumulated scar tissue impairs the liver's ability to filter toxins from the blood, leading to potential complications such as infections and liver cancer. While liver transplantation can be a life-saving intervention, there is a critical shortage of donor organs, resulting in approximately 1,000 deaths each year among patients awaiting transplants. Additionally, many patients are deemed ineligible for transplantation due to their deteriorating health conditions.
Injecting Hope: LyGenesis's Bold Procedure
The procedure devised by LyGenesis involves injecting healthy liver cells from a donor directly into a lymph node located in the upper abdomen of a patient with liver failure. The rationale behind this approach is that, over several months, these injected cells will proliferate and colonise the lymph node, forming a functional structure capable of performing the blood-filtering functions typically carried out by a healthy liver. This technique is seen as bold and innovative by experts like Valerie Gouon-Evans, a liver-regeneration specialist at Boston University, who commends the company's novel strategy.
Paving the way towards optimised organ donation
LyGenesis's innovative approach not only offers a potential solution for patients with liver failure but also aims to maximise the utilisation of donated livers that might otherwise go unused due to the lack of a suitable recipient within the transplant waiting list. The company's technique involves delivering donor cells through a tube inserted into the throat, targeting a lymph node near the liver for cell injection. Lymph nodes are considered ideal sites for growing 'mini livers' due to their abundant blood supply and the presence of numerous nodes throughout the body, allowing for the generation of these miniature organs without compromising the overall lymphatic system's function.
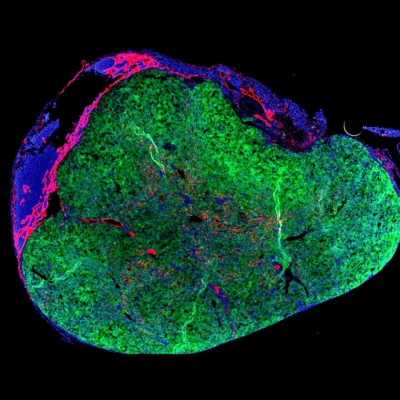
Preclinical success, from animal models to the first human patient
Promising results have been observed in preclinical studies involving mice, dogs, and pigs. In these animal models, miniature livers formed within two months following the injection of donor cells into lymph nodes, displaying a cellular structure resembling that of a healthy liver. Furthermore, these mini livers exhibited functional capabilities, including the transport of bile, a digestive fluid produced by the liver, without any signs of bile acid accumulation, indicating proper fluid processing. On March 25, the first human patient to undergo this experimental treatment was successfully administered the donor cells and is reportedly recovering well post-procedure. However, due to the introduction of foreign cells, the patient is required to take immunosuppressive drugs to prevent rejection of the donor cells, necessitating close monitoring for potential infections.
Human Trials: understanding the growth dynamics of Mini Liver
Michael Hufford, the CEO of LyGenesis, suggests that the growth of these mini organs is not indefinite and depends on distress signals emitted by the failing liver. Once these new organs establish blood-filtering capabilities, their growth will cease as the distress signals diminish. However, the exact size these mini-livers might reach in human patients remains uncertain. The company plans to enrol 12 participants in the phase II clinical trial by mid-2025, with results expected to be published the following year. The trial aims to evaluate participant safety, survival rates, and quality of life post-treatment, while also determining the optimal number of mini livers required to stabilise a patient's health. Clinicians conducting the trial will inject liver cells into up to five lymph nodes per participant to assess the procedure's efficacy in enhancing success rates.
Addressing Complications and Bridging Treatment Gaps
Despite the potential benefits offered by mini livers, they may not address all the complications associated with end-stage liver disease. For instance, portal hypertension, characterised by scar tissue-induced compression of liver blood vessels leading to internal bleeding, remains a significant concern. While mini livers may not alleviate this particular complication, they could serve as a temporary solution, bridging the gap until a suitable liver for transplantation becomes available or enabling patients to become healthier candidates for transplantation. Looking ahead, LyGenesis has broader ambitions beyond liver regeneration. The company is exploring similar approaches to cultivate kidney and pancreas cells within lymph nodes in animal models. If successful, this pioneering liver trial could pave the way for investigating the use of a patient's own stem cells to generate lymph node-seeding cells, potentially creating personalized cells that capture the liver's cellular diversity without the need for immunosuppressive drugs, as suggested by Gouon-Evans.
LyGenesis's experimental treatment for liver failure represents a groundbreaking approach that could revolutionise the treatment landscape for patients with end-stage liver disease. By harnessing the regenerative potential of donor liver cells and leveraging the body's lymphatic system, this innovative strategy offers a promising alternative to traditional liver transplantation, potentially saving lives and improving the quality of life for countless individuals worldwide.
Source & Image Credit: NATURE


![Tuberculosis Diagnostics: The Promise of [18F]FDT PET Imaging Tuberculosis Diagnostics: The Promise of [18F]FDT PET Imaging](https://res.cloudinary.com/healthmanagement-org/image/upload/c_thumb,f_auto,fl_lossy,h_184,q_90,w_500/v1721132076/cw/00127782_cw_image_wi_88cc5f34b1423cec414436d2748b40ce.webp)