HealthManagement, Volume 17 - Issue 2, 2017
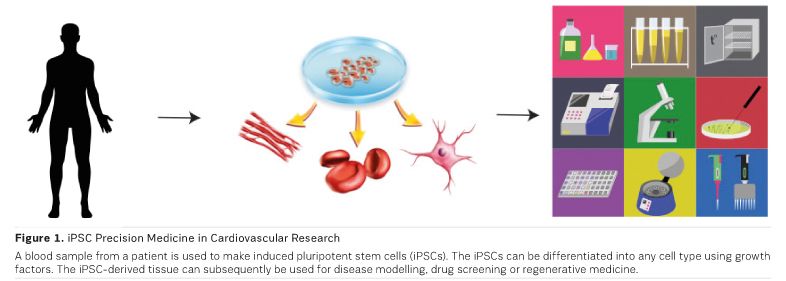
Induced pluripotent stem cells (iPSCs) are derived from somatic cells obtained from human subjects. iPSCs can be used for disease modelling, drug screening, and regenerative therapy. iPSCs have the potential to revolutionise patient care as part of precision medicine tailored to the individual.
Derived from terminally differentiated cells, iPSCs avoid the ethical problems of embryonic stem cells (ESCs). With patient consent, somatic cells can be obtained from a blood sample drawn from a vein to isolate peripheral blood mononuclear cells (PBMCs) or a skin sample that is used to isolate fibroblasts. Using a genetically engineered virus that overexpresses the transcription factors octamerbinding protein 3/4 (OCT 3/4; also known as PO U5F1), SRY (sex determining region Y)-box 2 (SOX 2), Myc proto-oncogene protein (c MYC), and Krüppel-like factor 4 (KLF4), the somatic cells are reprogrammed to become iPSCs (Takahashi et al. 2007). The iPSCs can then be differentiated into any tissue cell type using recombinant protein factors (Zhang et al. 2009). With this approach, iPSCs can be used to generate cardiovascular tissue to study complex disease states (Burridge et al. 2014; Orlova et al. 2014). iPSC disease modelling will vastly improve our ability to diagnose, prognosticate and treat cardiovascular disease.
Disease Modelling
iPSCs have advanced our understanding of the molecular mechanisms
of diseases such as dilated cardiomyopathy, hypertrophic cardiomyopathy, long
QT syndrome, and arrhythmogenic right ventricular cardiomyopathy (Kim et al. 2013;
Lan et al. 2013; Moretti et al. 2010; Sun et al. 2012; Wu et al. 2015). The
traditional method of studying human diseases uses animal models or
patient-derived tissue samples. Animal models, however, are problematic due to inherent
differences in physiology, reproducibility, ethical concerns and poor
correlation with human clinical trial data. Patient-derived tissue samples are
an excellent model for studying disease, but are limited in abundance and
availability. In contrast, iPS Cs can be differentiated into any tissue types,
including cardiac muscle cells and endothelial cells lining the inside of all blood
vessels. In contrast, iPSCs be used to identify genes responsible for a
disease. Subsequently, powerful new gene editing tools can be used to study
molecular mechanisms of disease (Hsu et al. 2014).
Uncovering disease
mechanisms can lead to the identification of new disease-specific biomarkers as
well as targets for new drug therapies (Mercola et al. 2013). Disease-specific
biomarkers can expedite the diagnosis of diseases and effectively predict their
responses to therapies, allowing for personalised treatment of each patient. Furthermore,
when mechanistic studies identify novel drug targets, patient-derived iPSCs can
be used to screen for beneficial versus toxic drug effects specific to the
genetic makeup of each patient. By contrast, it is not possible to predict
these effects accurately with prior approaches that use traditional animal
models.
Drug Screening
iPSC-derived tissues are unique to each individual. Not only can they be used to study individual susceptibilities to diseases, but they can also lead to tailored therapy, or personalised medicine. Patient-specific iPS C-derived tissue can be tested with conventional drug therapies to reveal the optimal treatment for each patient, avoiding a costly and inefficient trial-and-error approach that is prone to adverse reactions (Liang et al. 2013; Mathur et al. 2015). For example, the medical management of atrial fibrillation is complex. A doctor and patient discuss issues such as the risks and benefits of anticoagulation and, depending on symptoms, either a rate control or rhythm control strategy. Finding the optimal therapeutic regiment for a patient with atrial fibrillation, however, is challenging, time-consuming and can result in frequent emergency room visits, not to mention adverse side effects which are potentially life-threatening. By contrast, in the future, iPSCs from a patient with atrial fibrillation may be used to generate cardiomyocytes that could be safely tested with different rate and rhythm control agents. Using various in vitro assays, the optimal drug and dose could be determined empirically in a dish, thus avoiding drug titration and undesirable side effects.
The iPSC-derived tissues may also enhance clinical trials. For
example, a medication may not have an overall effect on a large population of
patients despite selection for patients with optimal demographics and risk factors.
Although a subpopulation may respond to a treatment, the effect is masked by
non-response in the larger heterogeneous population. To address this problem,
population-based studies using patient-derived iPSCs could preselect patients
with an optimal risk-to-benefit ratio prior to starting the clinical trial. A
rhythm control medication for atrial fibrillation, for instance, could be
tested in iPSC-cardiomyocytes from atrial fibrillation patients to predict who
would respond to a drug and also to test for adverse side effects such as drug-induced
arrhythmias. Patients identified to have safe and efficacious responses in the
iPSC screening would be subsequently enrolled into the trial. This enriched
sample population would have a larger chance of beneficial effect, which would
improve power and possibly reduce cohort size. If the drug were approved for
clinical use, an iPSC-cardiomyocyte screen could be used to predict which
patients in a general population would enjoy the greatest benefit for the new drug
in comparison to other antiarrhythmic agents.
See Also: Stem Cell Patch Improves Heart Failure Symptoms
Regenerative Medicine
Regenerative medicine is the holy grail of stem cell research. iPSCs represent a limitless source of tissue that could repair or replace organs (Lu et al. 2013). Because they are autologous, the use of iPS Cs could avoid immunosuppression that complicates standard organ transplants. Moreover, if the genetic cause of a patient’s disease is known, the mutation could be corrected with genome editing and a replacement organ could be synthesised in vitro and implanted. For example, patients with Marfan syndrome have a mutation in the fibrillin gene that causes valvulopathy and a dilated aorta. Over time, the valve and the aorta may need to be replaced with a prosthetic valve and aortic graft. With regenerative therapy using iPS Cs, the genetic mutation could be corrected with gene editing and a new aortic valve and aorta synthesised from iPSCs. However, there are several obstacles to translating regenerative therapies from the bench to clinical practice. From primary tissue collection to generating terminally differentiated iPS C-derived tissue, regenerative medicine requires an enormous amount of time and financial cost. Even under ideal conditions, the iPSC-derived tissues are notorious for batch to batch variability and immaturity. If regenerative therapy could overcome these hurdles, U.S. Food and Drug Administration approval would require that the iPSC-derived tissue be delivered in a targeted manner with no immunogenicity, tumorigenicity or toxicity (Neofytou et al. 2015). In addition, the transition to complex tissue and organ structure is challenging, since the three-dimensional structural organisation of tissues is lost in a petri dish. Nevertheless, advances in biomechanical engineering and emerging 3D printing technologies could make it possible to emulate the environmental configuration that facilitates the transformation of a complex cellular structure into an organ.
Conclusion
In summary, iPSCs are a powerful new modality in clinical medicine. They will not only enhance our understanding of diseases and lead to the discovery of new drug therapies, but will also advance personalised medicine and expedite clinical trials by enriching patient populations. The regenerative powers of iPS Cs have yet to be fully realised, but may one day lead to definitive treatments of end-organ dysfunction.
Key Points
- Induced pluripotent stem cells (iPSCs) are derived from somatic cells obtained from human subjects
- iPSCs can be used for disease modelling, drug screening, and regenerative therapy
-
iPSCs have the potential to revolutionise patient care as
part of precision medicine tailored to the individual
References:
Hsu PD, Lander ES, Zhang F (2014) Development and applications of CRISPR-Cas9 for genome engineering. Cell, 157(6): 1262-78.
Kim C, Wong J, Wen J et al. (2013) Studying arrhythmogenic right ventricular dysplasia with patient-specific iPSCs. Nature, 494(7435): 105-10.
Lan F, Lee AS, Liang P et al. (2013) Abnormal calcium handling properties underlie familial hypertrophic cardiomyopathy pathology in patient-specific induced pluripotent stem cells, Cell stem cell. 12(1): 101-13.
Liang P, Lan F, Lee AS et al. (2013) Drug screening using a library of human induced pluripotent stem cell-derived cardiomyocytes reveals disease-specific patterns of cardiotoxicity. Circulation, 127(16): 1677-91.
Lu TY, Lin B, Kim J et al. (2013) Repopulation of decellularized mouse heart with human induced pluripotent stem cell-derived cardiovascular progenitor cells, Nat Commun, 4: 2307.
Mathur A, Loskill P, Shao K et al. (2015) Human iPSC-based cardiac microphysiological system for drug screening applications. Sci Rep, 5: 8883.
Mercola M, Colas A, Willems E (2013) Induced pluripotent stem cells in cardiovascular drug discovery. Circ Res, 112(3): 534-48.
Moretti A, Bellin M, Welling A et al. (2010) Patient-specific induced pluripotent stem-cell models for long-QT syndrome. N Engl J Med, 363(15): 1397-409.
Neofytou E, O'Brien CG, Couture LA et al. (2015) Hurdles to clinical translation of human induced pluripotent stem cells. J Clin Invest, 125(7): 2551-7.
Orlova VV, van den Hil FE, Petrus-Reurer S et al. (2014) Generation, expansion and functional analysis of endothelial cells and pericytes derived from human pluripotent stem cells. Nat Protoc, 9(6): 1514-31.
Sun N, Yazawa M, Liu J et al. (2012) Patient-specific induced pluripotent stem cells as a model for familial dilated cardiomyopathy. Sci Transl Med, 4(130): 130ra47.
Takahashi K, Tanabe K, Ohnuki M et al. (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell, 131(5): 861-72.
Wu H, Lee J, Vincent LG et al. (2015) Epigenetic regulation of phosphodiesterases 2A and 3A underlies compromised beta-adrenergic signaling in an iPSC model of dilated cardiomyopathy. Cell Stem Cell, 17(1): 89-100.
Zhang J, Wilson GF, Soerens AG et al. (2009) Functional cardiomyocytes derived from human induced pluripotent stem cells. Circ Res, 104(4): e30-41.




