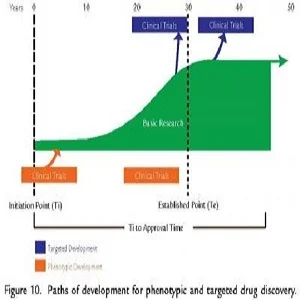
Death rates from cardiovascular disease (including heart disease and stroke) have declined markedly; still it remains the leading cause of death. Medical experts have expressed concern that the number of new therapies coming to market has lagged. New research suggests that the extensive delay in the emergence of new drugs reflects the long timelines for growth of basic research on cardiovascular disease.
The study, published in the journal Clinical Therapeutics, was a collaboration between the Center for Integration of Science and Industry at Bentley University and the Tufts Center for the Study of Drug Development at Tufts University School of Medicine. It traced the progress of developing new therapies, from basic science on mechanisms of cardiovascular disease through the development of new drugs based on this science.
Specifically, researchers tracked the development of 168 drugs currently approved for cardiovascular disease and 178 candidate drugs currently in development along with the maturation of the basic research underlying these products. They found that the median time from the initiation of research on new drug targets to first approval of a drug based on this research was greater than 40 years, with the time required for maturation of this research contributing the most to the delay in emergence of new products.
"The best way to speed drug development is to gain a better understanding of how basic research proceeds and how to accelerate this progress," said lead author Dr. Jennifer Beierlein.
"We have made a lot of progress in reducing regulatory review times and creating expedited paths to bring critical therapeutics to market more quickly," said Dr. Kenneth Kaitin, Professor and Director at the Tufts Center for the Study of Drug Development. "By looking more holistically at the innovation pathway, this research tells us what we need to do next to accelerate development even further."
This study highlights the critical importance of continued support for the basic research required to discover and develop the next generation of drugs for cardiovascular disease. The authors also point to the risk of initiating drug development without a sufficiently established basis of scientific research.
Source: Bentley University
Image Credit: Center for Integration of Science and Industry
References:
Beierlein, Jennifer M. et al. (2017) Landscape of Innovation for Cardiovascular Pharmaceuticals: From Basic Science to New Molecular Entities. Clinical Therapeutics; DOI: 10.1016/j.clinthera.2017.06.001
Latest Articles
heart disease, new drugs, emergence of new drugs, scientific research
New research suggests that the extensive delay in the emergence of new drugs reflects the long timelines for growth of basic research on cardiovascular disease.