A novel way to image the entire brain’s glymphatic pathway, a dynamic process that clears waste and solutes from the brain that otherwise might build up and contribute to the development of Alzheimer’s disease, may provide the basis for a new strategy to evaluate disease susceptibility, according to a research paper published online in The Journal of Clinical Investigation. Through contrast enhanced magnetic resonance imaging (MRI) and other tools, a Stony Brook University-led research team successfully mapped this brain-wide pathway and identified key anatomical clearance routes of brain waste.
In their article titled “Brain-wide pathway for waste clearance captured by contrast enhanced MRI,” Principal Investigator Helene Benveniste, MD, PhD, a Professor in the Departments of Anesthesiology and Radiology at Stony Brook University School of Medicine, and colleagues built upon a previous finding by Jeffrey Iliff, PhD, and Maiken Nedergaard, MD, PhD, from University of Rochester that initially discovered and defined the glymphatic pathway, where cerebral spinal fluid (CSF) filters through the brain and exchanges with interstitial fluid (ISF) to clear waste, similar to the way lymphatic vessels clear waste from other organs of the body. Despite the discovery of the glymphatic pathway, researchers could not visualise the brain wide flow of this pathway with previous imaging techniques.
“Our experiments showed proof of concept that the glymphatic pathway function can be measured using a simple and clinically relevant imaging technique,” said Dr. Benveniste. “This technique provides a three-dimensional view of the glymphatic pathway that captures movement of waste and solutes in real time. This will help us to define the role of the pathway in clearing matter such as amyloid beta and tau proteins, which affect brain processes if they build up.”
Dr. Benveniste said that the pathology of certain neurological conditions is associated with the accumulation of these proteins and other large extracellular aggregates. In particular, she explained that plaque deposits of these proteins are implicated in the development of Alzheimer’s disease, as well as chronic traumatic encephalopathy that occurs after repetitive mild traumatic brain injuries.
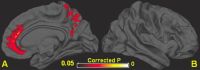
The researchers used contrast agents and molecular tracers with MRI to develop the imaging tools. Using these tools, they imaged major regions within the brain such as the cerebellum, orbito-frontal cortex, pineal gland, olfactory bulb, and nasal cavity, to map the entire glymphatic pathway. The contrast-enhanced images reveal the CSF-ISF interchange throughout the whole brain, including pathways in parallel to major arteries that are also involved in the glymphatic waste clearing process.
This advanced imaging technique has the potential to be used as a way to monitor the human brain to map brain waste clearance and assess disease susceptibility. Dr. Benveniste pointed out that, theoretically, if clinicians were able to capture a defect in the glymphatic system where certain channels are malfunctioning, plaque formation would likely accelerate. This plaque buildup may be an early sign of disease susceptibility before evidence of any cognitive changes. Though there is no known way to repair malfunctions in the glymphatic system, the research team is investigating ways to repair or open malfunctioning channels.
Co-authors include Jeffrey J. Lliff and Maiken Nedergaard of the Center for Translational Neuromedicine at the University of Rochester in Rochester, N.Y.; Jean Logan of Brookhaven National Laboratory and the Department of Radiology, Stony Brook Medicine; Hedok Lee and Mei Yu of the Department of Anesthesiology, Stony Brook Medicine; and Tian Feng of the Department of Applied Mathematics and Statistics, Stony Brook University.
Reference: Jeffrey J. Iliff, Hedok Lee, Mei Yu, Tian Feng, Jean Logan, Maiken Nedergaard and Helene Benveniste. "Brain-wide pathway for waste clearance captured by contrast-enhanced MRI" J Clin Invest. doi:10.1172/JCI67677.
Latest Articles
Research, Brain
A novel way to image the entire brain’s glymphatic pathway, a dynamic process that clears waste and solutes from the brain that otherwise might build up...