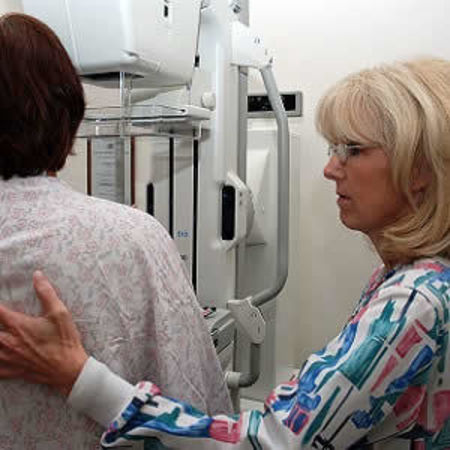
A new article supports the recent changes made by the American Cancer Society in its recommendations for breast cancer screening. The new recommendations have modified the suggested ages for annual and biannual mammography and have also focused on patient preferences in decision making.
The three primary recommendations by the ASC included:
1. Annual screening to begin at age 45 with no objections to begin as early as 40.
2. Biannual screening starting at age 55 and the option to continue with annual screening if preferred.
3. Discontinued use of clinical breast examinations as a screening mechanism for women at any age.
When it comes to breast cancer screening practices, it is important to have a delicate balance between the dangers of insufficient monitoring and unnecessary testing. The primary dangers of insufficient monitoring are failing to know if a woman has cancer and delaying treatment while the dangers of unnecessary testing include an increased exposure to radiation and unnecessary false positives.
The article talks about the important differences between the sensitivity and false positive rates for mammography. While mammography detects 84 percent of breast cancers in asymptomatic women, nearly 95 percent of all positive mammograms are false positives.
"Our goal was to caution asymptomatic women that positive mammograms are vastly more likely to be false positives than actual evidence of cancer," said Dr. William Skorupski, co-author of the Significance article. "For most women, a false positive mammogram is about 19 times more likely than a true positive," added co-author Dr. Howard Wainer.
Source: Wiley
Image Credit: Wikimedia Commons
Latest Articles
breast cancer screening, American Cancer Society, mammography, radiation
A new article supports the recent changes made by the American Cancer Society in its recommendations for breast cancer screening. The new recommendations have modified the suggested ages for annual and biannual mammography and have also focused on patient